In the field of tumor therapy driven by nanozymes, precise regulation of the electronic states of nanomaterials is crucial for interfering with the redox homeostasis within cancer cells. Rare earth elements, due to their unique 4f orbital electronic configuration, have emerged as potential highly effective antitumor catalysts. However, the strong localization, significant shielding effect, and spin-orbit coupling characteristics of 4f electrons make it difficult to effectively regulate their electronic states through traditional methods, severely limiting the enhancement of their catalytic activity and the expansion of biomedical applications.
Amorphous materials, characterized by their "short-range order and long-range disorder" structure, are rich in structural defects and unsaturated coordination sites. This can enhance the orbital overlap and hybridization between 4f orbitals, thereby achieving dynamic regulation of the electronic states of rare earth elements and improving their catalytic activity. This provides a novel approach for the development of efficient anti-tumor nanozymes.
Addressing the aforementioned scientific challenges, the authors designed and synthesized an amorphous cage-like Gd(OH)3 nanozyme based on the "interface-confined coordination etching" strategy first proposed by Professor Guo Lin's team in 2013 (J. Am. Chem. Soc., 2013, 135, 16082). The amorphization significantly reduces the coordination number of Gd-O and alters the 4f electronic state, enhancing the delocalization of holes. This allows the originally inert Gd3+ (with a half-filled 4f7 orbital) to exhibit excellent peroxidase (POD)-like activity, with a Kcat of 3.49×10⁴ s-1, which is an order of magnitude higher than that of the natural HRP enzyme. After PEG modification, the nanocage can be specifically activated in the tumor microenvironment (pH ≈ 5.5) and inhibits tumor growth by efficiently generating reactive oxygen species (ROS). This demonstrates excellent therapeutic potential and provides a new direction for the application of rare earth nanomaterials in the biomedical field.
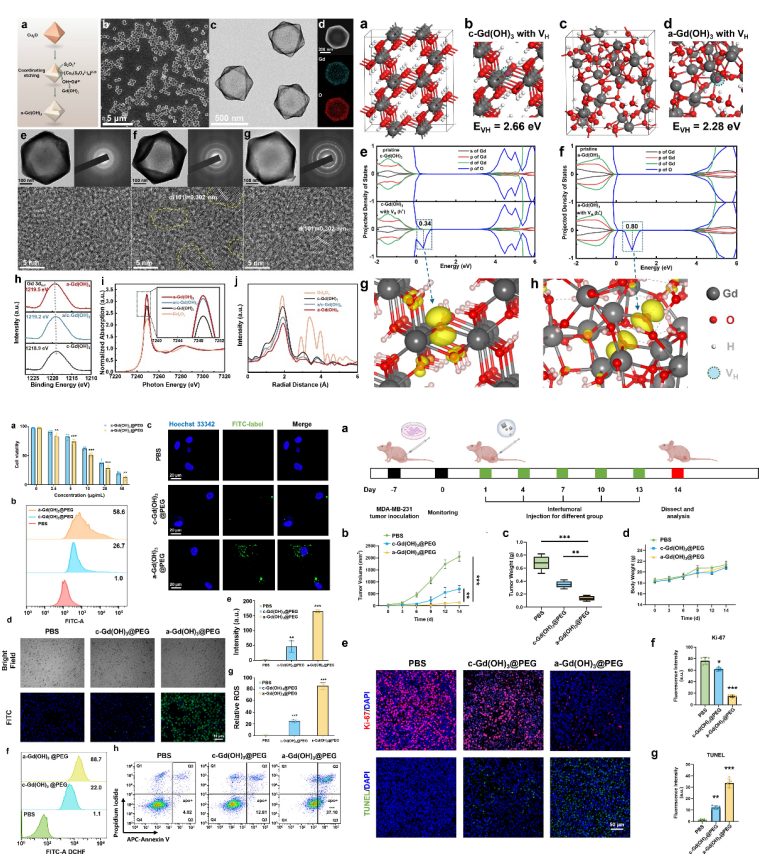
Zezhou Wang, Qi Hu, Mengmeng Zhang, Chen Li, Shu Wang, Yanhong Li, Fengshi Li, Tianqi Guo*, Pengfei Hu*, Kewei Jiang*, Gilberto Teobaldi, Li-Min Liu*, and Lin Guo* Amorphization-Induced Electronic Modulation of Gd (OH)3 Nanocages with Enhanced Enzymatic Activities for Antitumor Therapy. Journal of the American Chemical Society 2025.
https://doi.org/10.1021/jacs.5c09591