The catalytic performance of rare earth elements is highly dependent on their 4f electronic states. However, the regulation of 4f electrons has long faced multiple challenges: the 4f orbitals are located in the inner shell of the atom and are strongly shielded by the outer 5s and 5p electrons, making it difficult for them to effectively couple with the external environment; their electron cloud distribution is highly localized, and the ordered lattice structure of crystalline materials strictly limits the dynamic behavior of 4f electrons. Traditional crystal doping or defect engineering is difficult to break this localization. In addition, high concentrations of Ce3+ are prone to induce phase transitions (such as CeO2→Ce2O3), resulting in material deactivation.
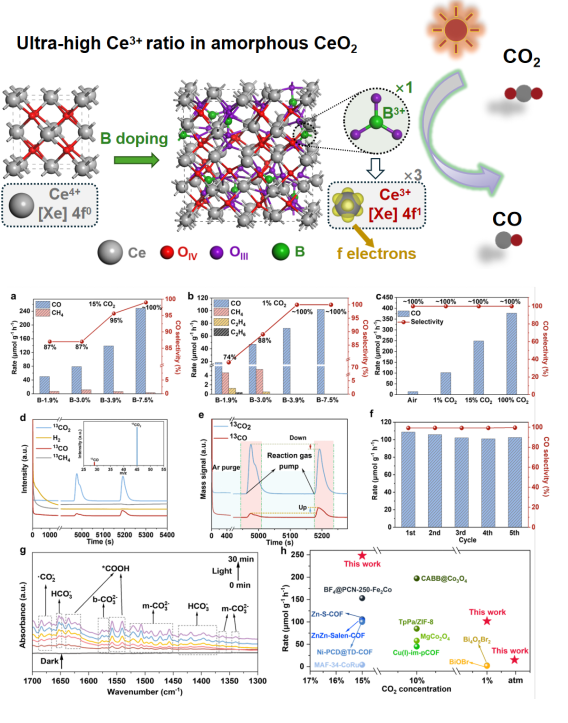
Addressing the challenging issue of regulating 4f electrons in rare earth elements, the authors propose an innovative strategy: inducing amorphous transformation of CeO2 through hetero-ion doping, breaking the constraint of crystal symmetry on 4f electrons, and achieving effective regulation of 4f electron states and significant enhancement of Ce3+ content. The key to this amorphous transformation strategy lies in disrupting the ordered structure of symmetry in traditional CeO2 crystals. The research team introduced B doping, allowing each B atom to bond with three O atoms, forming a planar BO33- group. This structural unit not only weakens the original Ce–O bond, inducing the formation of a large number of three-coordinated oxygen (OIII) sites, but also disrupts the crystal symmetry, successfully promoting the amorphous transformation of CeO2. By regulating the content of B doping (1.9% to 7.5%), precise control over the degree of amorphous transformation of CeO2/GO nanosheets can be achieved. As the B doping content increases, the proportion of Ce3+ continues to rise, and the results are highly consistent with theoretical predictions, indicating that this amorphous transformation strategy effectively broadens the range of electronic state regulation. In low-concentration CO2 photocatalytic reduction tests, the B-7.5% catalyst exhibited excellent performance: at a CO2 concentration of 15%, the CO generation rate reached 249.33 μmol·g-1·h-1 with a selectivity of 100%; even when the CO2 concentration was reduced to 1%, the rate remained at 103.4 μmol·g-1·h-1, far exceeding similar catalysts.
This study systematically verifies for the first time the feasibility of the amorphous strategy for regulating the electronic states of rare earth 4f electrons, breaking through the symmetry constraints on electronic states in traditional crystal structures and significantly expanding the regulation range of 4f electrons. This strategy not only achieves efficient photocatalytic reduction of CO2 under low concentration conditions but also provides a new approach for the structural design and performance optimization of rare earth nanomaterials.
Y. Li, Q.Yin, B.Jia, H.Wang, H.Gu, Q.Hu, H.Yang, T.Guo, P.Hu, L.Li, L.-M.Liu, L.Guo, Angew. Chem. Int. Ed.2025, 64, e202505668.
https://doi.org/10.1002/anie.202505668