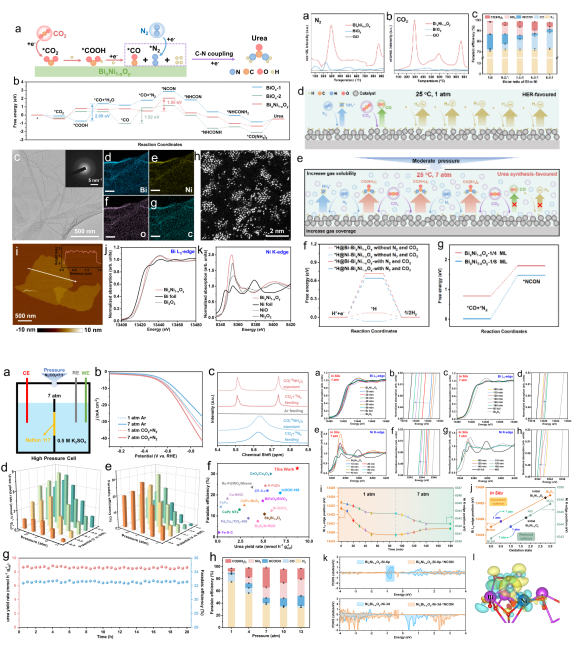
Although electrochemical nitrogen fixation technology can achieve the conversion of nitrogen, the activation process of nitrogen in aqueous systems is dominated by competitive hydrogen evolution side reactions, resulting in low yield and Faraday efficiency. Inspired by Le Chatelier's principle, this study innovatively introduces a pressure field into the electrochemical system for the co-electrolysis of N2 and CO2 to synthesize urea. By applying moderate pressure, it suppresses gaseous by-products such as CO and H2 while promoting C-N coupling. The study designs an atomically dispersed amorphous BixNi1-xOy cluster catalyst, confirming that pressure-driven changes in catalyst surface species coverage trigger in-situ electronic structure regulation: the oxidation state of Bi species gradually increases, while that of Ni species gradually decreases. This atomically coordinated design of multi-metal active centers, combined with a systematic engineering strategy of regulating the interfacial microenvironment through moderate pressure, achieves breakthrough catalytic performance: a urea yield of 8.71 mmol h-1 g-1cat and a nitrogen fixation efficiency of up to 50%. This work, through the integration of pressure engineering and atomically designed catalysts, provides a reference for gas-involved electrochemical reactions and technical support for the artificial closed-loop of carbon and nitrogen cycles in nature.
Xiangyu Chen, Tongcai Yue, Yue Liu, Yong Zhao, Hanke Cui, Hongfei Gu, Xiaoyi Sun, Qi Hu, Mingke Sun, Haolin Li, Yuhan Ma, Weifeng Huang*, Jianxin Kang*, Li-Min Liu*, Lin Guo*. Efficient Electrochemical Urea Synthesis From CO2 and N2 in Moderate Pressure. Angewandte Chemie International Edition, 2026.
https://onlinelibrary.wiley.com/doi/10.1002/anie.8253479